Know Your Battery
HOW BATTERY WORKS
An elaborate electrical system of circuits within an automobile is needed just to produce, store, and distribute all the electricity that it requires for its daily operations. The battery is the first major component in a car's power system. It is used for storing power for ignition, and for running auxiliary devices such as onboard computers, clocks, radios and alarms when the engine is off.
When a car's ignition is turned "ON", a signal is sent to the car's battery. Upon receiving this signal, the battery releases the energy (stored in the form of chemical energy) as electricity, which in turn is used to crank the engine. The battery also releases energy to ignite the fuel and meet other demands not met by the alternator.
HOW TO JUMP START A VEHICLE
Vehicle Jump-Starting procedures as listed below should be followed carefully:
• Ensure that vehicle battery and battery for jump-starting have the same declared voltage (say 12V)
• Turn off all electrical loads, check vehicles - no part should be touching each other and gear should be in neutral position
• Vehicles should have the same terminal earthed, if not, refer to manufacturers' instructions. Make sure that cables are not frayed or damaged.
Carry out the following steps in sequence:
• Connect the positive terminal of jump-starting battery to the positive terminal of the stalled car battery - on negative grounded system.
• Connect one end of the other cable to the negative (-) terminal of the booster battery.
• Connect the other cable away from the battery, to the engine block or car frame of the vehicle to be started.
• Make sure cables are away from fan blades and other moving vehicle parts.
• After starting, remove cables in reverse order, starting with one connected to engine block or car frame.
• If vehicle does not start within 30 seconds, call an auto electrician.
Caution:
Modern vehicles with electronic management systems should not be jump-started without "protected" jump starter leads. It is necessary to refer to the owner's handbook for jump-starting procedures for such vehicles.
BATTERY DEFINITIONS
Acid: Refers to sulphuric acid (H2SO4), or a mixture of sulphuric acid and water - the active component of the electrolyte.
Active Material: Indicates the active portion of the battery plates; peroxide of lead on the positives and spongy metallic lead on the negative.
Alternating Current: Electric current, which unlike direct current, rapidly reverses its direction or "alternates" in polarity so that it doesn't charge a battery.
Ampere: The unit that measures the rate of flow of electric current.
Ampere Hour: It is the amount of energy charge in a battery that will allow one ampere of current to flow for one hour.
Battery: A device that comprises a group of electric cells that converts chemical energy into electrical energy to act as a source of direct current
Buckling: Warping or bending of the battery plates.
Capacity: The number of ampere-hours that a battery can supply at a given rate of current flow after being fully charged. e.g., a battery may be capable of supplying 8 amperes of current for 10 hours before it is exhausted. Its capacity is 80-ampere hours at the 10 hours rate of current flow. It is necessary to state the rate of flow, since same battery if discharged at 20 amperes would not last for 4 hours but for a shorter period, say 3 hours. Hence, its capacity at the 3 hour rate would be 3x20=60 ampere hours.
Case: The container that holds the battery cells.
Cell: The battery unit that comprises an element complete with electrolyte.
Charge: Passing direct current through a battery in the direction opposite to that of discharge, in order to restore the energy used on discharge.
Charge Rate: The rate of current that is required for charging a battery from an external source. The rate is measured in amperes and varies for cells of different size.
Corrosion: It is the erosion of metal parts on exposure by acid from the electrolyte.
Cycle: One discharge and charge.
Density: Specific gravity
Discharge: The flow of current from a battery through a circuit, opposite of "charge."
Dry: Term frequently applied to cell containing insufficient electrolyte. Also applied to certain conditions of shipment of batteries (i.e. without electrolyte)
Electrolyte: The conducting fluid of electro-chemical devices (for lead-acid storage batteries electrolyte comprises about two parts of water to one part of chemically pure sulphuric acid, by weight)
Element: Positive group, negative group and separators.
Evaporation: Loss of water from electrolyte due to heat or charging.
Filling Plug: The plug that fits in and closes the orifice of the filling hollow in the cell cover.
Freshening Charge: A charge given to an idle battery to keep it fully charged.
Gassing: Emission of oxygen at the positive plates and hydrogen at negatives, which begins when charge is nearing completion.
Gravity: A contraction of the term "specific gravity," which means the density compared to water as a standard.
Grid: The metal framework of a plate that supports the active material and is provided with a lug for conducting the current.
Group: A set of plates, either positive or negative, joined to a strap. Groups do not include separators.
Hold-Down: Device to keep separators in place.
Hold-Down Clips: Brackets for the attachment of bolts for holding the battery securely in position within the car.
Hydrometer: An instrument for measuring the specific gravity of the electrolyte.
Hydrometer Syringe: A glass barrel enclosing a hydrometer and provided with a rubber bulb for drawing up electrolyte.
Lug: The extension from the top frame of each plate, connecting the plate to the strap.
Maximum Gravity: The highest specific gravity to which the electrolyte will reach by continued charging, indicating that no acid remains in the plates.
Negative: The terminal of a source of electrical energy as a cell, battery or generator through which current returns to complete circuit. Generally marked "Neg."
Over-Discharge: The carrying of discharge beyond proper cell voltage; this activity shortens battery life, if carried far beyond proper cell voltage and done frequently.
Paste: The mixture of lead oxide or spongy lead and other substances, which is put into grids.
Plate: The combination of properly "formed" grid and paste. Positive are reddish brown and negatives slate gray.
Polarity: A battery has two poles with opposing attributes. The positive terminal (or pole) of a cell or battery or electrical circuit is said to have positive polarity; the negative terminal has negative polarity.
Hydrometer: The terminal of a source of electrical energy as a cell, battery or generator from which the current flows. It is generally marked "Pos.".
Post: The portion of the strap extending through the cell cover, by means of which connection is made to the adjoining cell or to the car circuit.
Potential Difference: Abbreviated P. D. and found on test curves. The term is synonymous with voltage.
Rate: Number of amperes (rate of flow of electricity) for charge or discharge. Also used to express time for either.
Reversal: Reversal of polarity of cell or battery, due to excessive discharge, or charging in the wrong direction.
Separator: An insulator between plates of opposite polarity; usually of wood, rubber or a combination of both. Separators are generally corrugated or ribbed to insure proper distance between plates and to avoid too much displacement of electrolyte.
Short Circuit: A low-resistance connection between two points in an electric circuit. Short circuit happens when the current tends to flow through the area of low resistance, bypassing the rest of the circuit.
Specific Gravity: The density of the electrolyte compared to water as a standard. It indicates the strength of the electrolyte and is measured by the hydrometer.
Strap: The leaden casting to which the plates of a group are joined.
Sulphate: Common term for lead sulphate (PbSO4.)
Sulphated: Term used to describe cells in an under-charged condition, from either over- discharging without corresponding long charges or from remaining idle for a specific time and being self-discharged.
Terminal: It is the electrical connection from the battery to the external circuit. Each terminal is connected to either the positive (first strap) or negative (last strap) in the series connection of cells in a battery.
Vent Plug or Vent-Cap: Hard or soft rubber/ plastic part inserted in cover to retain atmospheric pressure within the cell, while preventing loss of electrolyte from spray. It allows gases formed in the cell to escape, prevents electrolyte from spilling, and keeps dirt out of the cell.
Volt: It is the electrical unit of pressure in an electric circuit. Voltage is measured by a voltmeter. It is analogous to pressure or head of water flow through pipes. NOTE. - Just as increase of pressure causes more volume of water to flow through a given pipe, so increase of voltage (by putting more cells in circuit) will cause more amperes of current to flow in the same circuit. Decreasing size of pipes increases resistance and decreases flow of water. Introduction of resistance in an electrical circuit decreases current flow with a given voltage or pressure.
BATTERY MAINTENANCE
• Acid: The maintenance of auto battery involves periodic checking of the battery so that your car runs smoothly. Keep in mind the following for a longer hassle-free battery life:
• Check Clamp: Make sure that the battery is firmly secured to the cradle and the cable clamps and lead wire contact is proper.
• Avoid Grease: Keep the battery top clean and dry. Apply either petroleum jelly or Vaseline to cable clamps and terminals for proper lubrication. Never apply grease.
• Use Distilled Water: Top up only with distilled water and maintain the level to the line that indicates, maximum. Never add acid.
• Close Tightly: Keep the vent plugs closed tightly.
• Check Vent: Ensure that the vent hose in the battery is not folded or damaged by the exhaust system.
• Check Regularly: Be sure to inspect your vehicle's electrical system regularly, especially the regulator voltage setting
• Service Regularly: Get your battery serviced regularly from your nearest authorised Exide dealer.
BATTERY HANDLING
 It is extremely important to be cautious while handling a battery, as it can cause several hazards. Some of the possible risks are mentioned below to direct motorists to handle car batteries with utmost care:
It is extremely important to be cautious while handling a battery, as it can cause several hazards. Some of the possible risks are mentioned below to direct motorists to handle car batteries with utmost care:
Battery Acid can cause burns:
Be extremely careful while handling battery acid. If electrolyte is spilled on clothing or skin, wash with plain water and neutralise with a solution of baking soda and water. Electrolyte splashed into eyes can cause serious damage. In the event of such an accident, splash affected eye with clean and cold water for at least 5 minutes. Take medical advice.
Handling the Electrolyte:
(a) Preparing electrolyte:
In case electrolyte needs to be prepared, always add concentrated acid to water and never water to acid, as it may cause effervescence and spillage. Store electrolyte in plastic containers with sealed cover. Keep away from direct sunlight.
(b) In case electrolyte is swallowed:
Drink large quantities of water or milk and follow with milk of magnesia, beaten eggs or vegetable oil. Take medical advice.
Battery Explosion:
Batteries generate explosive gases during vehicle operation and also when charged separately. Flames, sparks, burning cigarettes or other inflammable sources that may start a fire must be kept away at all times.
Caution
• Always shield eyes when working with batteries, as sparks, acid splurge etc. may hurt the eyes.
• When charging batteries, work in a well-ventilated area - never in closed spaces
• Always turn battery charger or ignition off before disconnecting a battery.
BATTERY RECYCLING:
Don't let your used battery fall into the wrong hands
Lead, a major component in all batteries, if not handled with care after use, can become hazardous and seriously harm the environment and the people around. When smelted in open furnaces, by unauthorised smelters, scrap lead particles escape into the air. Unchecked, they infect workers, permeate the soil and contaminate groundwater, pollute all organic life: vegetation, fish, meat, poultry, livestock and human beings in the process.
Even a slight exposure to lead can cause severe kidney damage, cerebro-muscular failure, a slowing down of the nervous system, abdominal discomfort, anaemia, hypertension and other major diseases. In areas exposed to open lead furnaces, there have even been cases of spontaneous abortion.
How lead pollution takes place through unauthorised smelting
• Lead particles settling on the floor stick to clothes or are swept into dust
• Lead is released in workplace atmosphere as dust or fumes
• Minute lead particles are ingested with food and drink
• Air borne particles settle on workmen's skin / clothing
• Lead particles being heavier enter soil and sub-soil
• Lead, inhaled by workmen, settles in the lungs
Save your children and the planet from lead poisoning
Handle used batteries with care. As responsible citizens, keep the planet and the generations to come free of lead poisoning. We at Exide collect used batteries at regular intervals from our dealers in closed trucks and have them recycled by smelters, authorised by the Ministry of Environment. We encourage dealers to collect all batteries that have lived out their service life.
All you need to do is return your used battery to any of our authorised dealers. This will ensure that your used battery never pollutes the environment or endangers the health of your children. Let us join hands to keep life on earth clean, beautiful and sustainable.
Caution
• Always shield eyes when working with batteries, as sparks, acid splurge etc. may hurt the eyes.
• When charging batteries, work in a well-ventilated area - never in closed spaces
• Always turn battery charger or ignition off before disconnecting a battery.
Lead, one of the most useful metals, if not treated and preserved after use, can turn hazardous.
Avail of a special rebate on Exide batteries every time you return a used battery.
Return used batteries. Protect the environment.
BATTERY FAQS
A. WHAT ARE THE FEATURES THAT SHOULD BE CONSIDERED WHEN BUYING A BATTERY?
Size: The dimensions of your original battery
Power: The Ampere hour required to power your vehicle
Warranty: Automotive batteries are backed by a warranty package. Choose the one that is right for your vehicle's needs.
B. WHILE REPLACING THE BATTERY OR CLEANING THE TERMINALS, WHY IS IT IMPORTANT TO REMOVE THE GROUND WIRE FIRST?
Before you start, always check the type of grounding system the vehicle has. If you remove the positive connector first in a negative ground system, a spark may be created in the process. It could happen if the metal tool you're using to remove the positive terminal connector comes in contact with any piece of metal in the car. If you are working near the battery when this occurs, it might create fire source causing the battery to explode. It's extremely important to remove the ground source first.
C. WHAT CAN EXCESSIVE HEAT DO TO A BATTERY?
Heat can deteriorate a battery's life quicker by evaporating the water from the electrolyte, and corroding and weakening the positive grids.
D. IF MY CAR DOESN'T START, HOW DO I KNOW FOR SURE THAT MY BATTERY NEEDS REPLACEMENT?
Many other problems can keep a car from starting. It is advisable to do some troubleshooting. Exide authorised dealers test batteries free of charge, so as a good first step visit your nearest Exide Dealer to be sure of your battery.
E. HOW DO I SAFELY JUMP-START MY BATTERY?
Check out "How to jump start a vehicle"
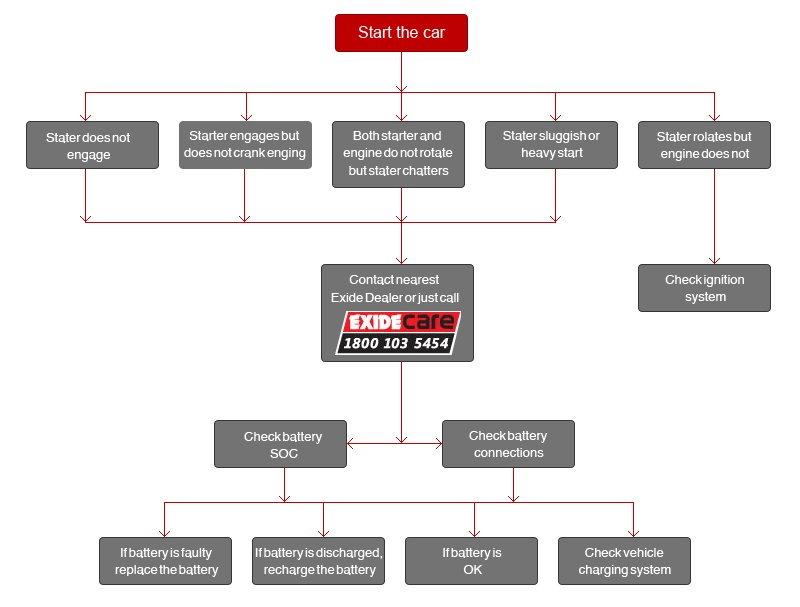
HOW TO KEEP YOUR CAR OUT OF TROUBLE
Facing a starting problem? Follow these steps!
When you attempt to start your car, you may be faced with two fold problems -
a) The car engine turns well but does not start
b) The engine itself does not turn or turns very slowly.
In case of (a) you need to check the engine, carburetor, ignition system etc.
In case of (b) if the weather is cold, the engine may take time to warm up. Be sure to check the grade of oil once. Or else, check your battery connection - in case of any problem, contact your nearest Exide dealer or Bat Mobile Service. Replace your vehicle battery, if it is faulty or recharge if it is low on charge and check fan belt and alternator output. Don't forget to test operating conditions. However, if battery function is smooth, check the starter motor and its connections.
Take a tour - step by step - and drive your car out of trouble.